 Autism research continues to emerge at a steady pace. “A new, integrated interpretation of Autism Spectrum Disorders (ASD)” was recently reported. Now, researchers have found a potential biomarker for Autism as, for the first time, inherited disruption of gene expression in a brain system for social behavior has been implicated in autism. National Institute of Mental Health (NIMH) grantee Margaret Pericak-Vance, Ph.D., at the University of Miami and Simon Gregory, Ph.D., at Duke University, and a multinational team of researchers found evidence for such epigenetic effects on the gene for the oxytocin receptor – part of a brain system that mediates social behaviors disturbed in autism. The findings suggest a potential genetic biomarker for the disorder. The researchers report on their findings online October 22, 2009 in BMC Medicine.
Autism research continues to emerge at a steady pace. “A new, integrated interpretation of Autism Spectrum Disorders (ASD)” was recently reported. Now, researchers have found a potential biomarker for Autism as, for the first time, inherited disruption of gene expression in a brain system for social behavior has been implicated in autism. National Institute of Mental Health (NIMH) grantee Margaret Pericak-Vance, Ph.D., at the University of Miami and Simon Gregory, Ph.D., at Duke University, and a multinational team of researchers found evidence for such epigenetic effects on the gene for the oxytocin receptor – part of a brain system that mediates social behaviors disturbed in autism. The findings suggest a potential genetic biomarker for the disorder. The researchers report on their findings online October 22, 2009 in BMC Medicine.
Background
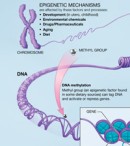
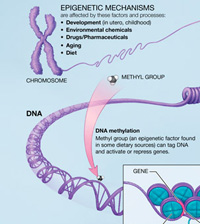
Environmentally-influenced and heritable chemical tags called epigenetic marks regulate the mix of proteins needed to build each tissue of the body. These alterations do not change a person’s DNA, or genetic blueprint. However, epigenetic changes that occur from the moment sperm meets egg can alter when and where genes get turned on.
Researchers are testing oxytocin as a possible treatment for social behavior disturbances in autism. Oxytocin is a hormone produced in the brain, which works through the oxytocin receptor. Previous studies had hinted at oxytocin receptor abnormalities in autism, but not at an epigenetic mechanism.
The researchers first used high-tech, genome-wide techniques to search for deletions or duplications of genes in 119 people with autism from families that had multiple children with the disorder and 54 healthy controls. They then searched in brain tissue and blood samples from affected and unaffected individuals for a common type of epigenetic mark created by methylation. In this process, molecules called methyl groups attach to DNA in response to an environmental trigger, preventing a gene’s expression.
Findings of This Study
The researchers found a deletion in the oxytocin receptor gene in a person with autism and his mother, who had obsessive compulsive disorder (OCD). OCD shares with autism symptoms of repetitive behaviors. Oxytocin receptor genes were similarly silenced – but by methylation – in a sibling with autism who lacked the deletion. That is, two separate mechanisms of gene expression regulation resulted in the same outcome – loss of oxytocin receptor expression – in the same family.
Following up in blood cells and temporal cortex brain tissue of people with autism, the researchers pinpointed higher levels of methylation – about 70 percent vs. the normal 40 percent – at an epigenomic site known to regulate the oxytocin receptor. They also found decreased expression of the receptor in the temporal cortex tissue, an area previously linked to autism.
Significance
Excess methylation of the oxytocin receptor could render people with autism less sensitive to the social hormone’s effects. Gene expression most likely became altered in very early gestation (between fertilization and implantation), suggest the researchers. This could increase vulnerability of the oxytocin receptor gene to environmental insults during the first few weeks of pregnancy, they say. The results suggest that such epigenetic misregulation of the oxytocin receptor gene may be an important factor in the development of autism.
What’s Next?
Since evidence of excess methylation of the oxytocin receptor gene in temporal cortex was also found in blood cells, the researchers suggest that the blood measure may be a marker, more generally, of the methylation status of the temporal cortex. So measuring the methylation status of the oxytocin receptor in blood could potentially serve as a biomarker for autism that might be used in conjunction with traditional diagnostic criteria. Drugs that target methylation might also hold promise for treatment.
Material adapted from National Institute of Mental Health by CFisher.
Reference
Genomic and epigenetic evidence for oxytocin receptor deficiency in autism. Gregory SG, Connelly JJ, Towers AJ, Johnson J, Biscocho D, Markunas CA, Lintas C, Abramson RK, Wright HH, Ellis P, Langford CF, Worley G, Delong GR, Murphy SK, Cuccaro ML, Persico A, Pericak-Vance MA. BMC Med. 2009 Oct 22;7(1):62. [Epub ahead of print]PMID: 19845972.
No comments yet.